|
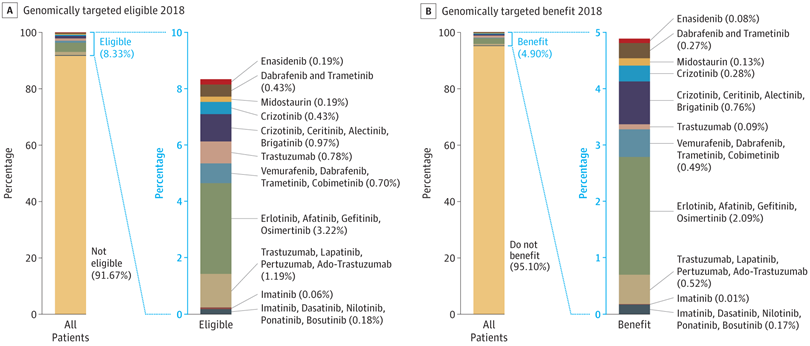
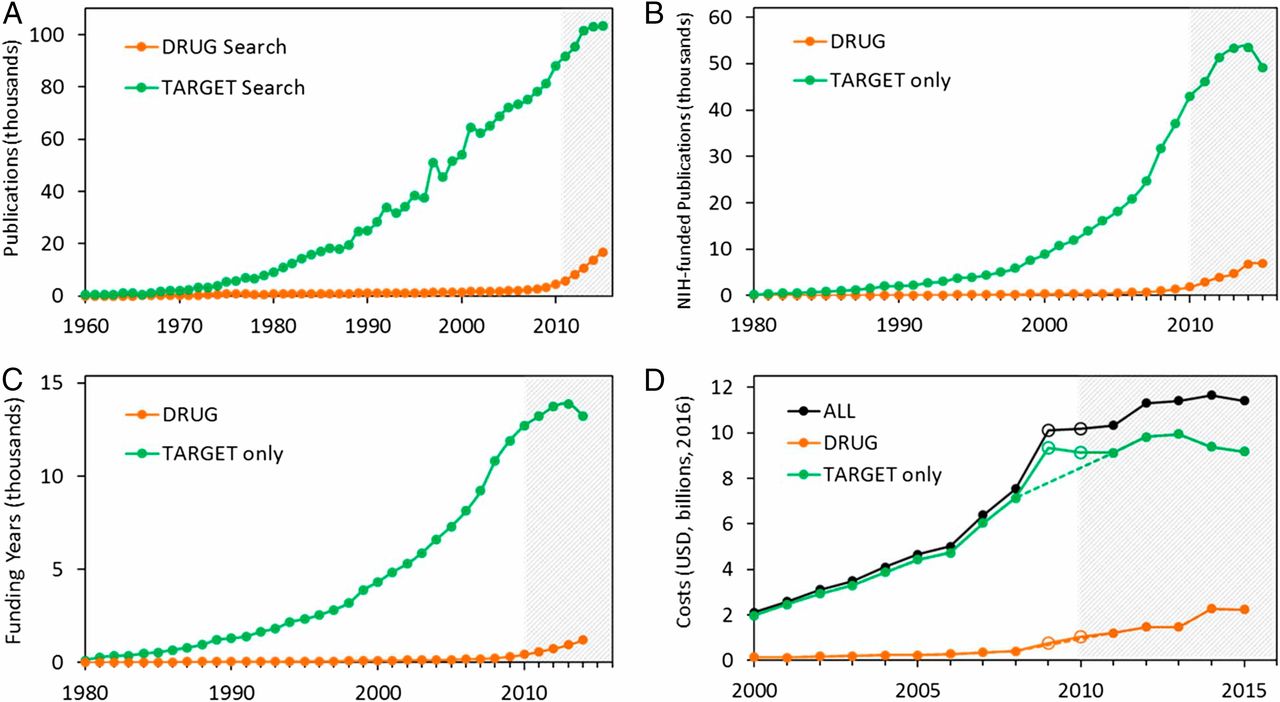
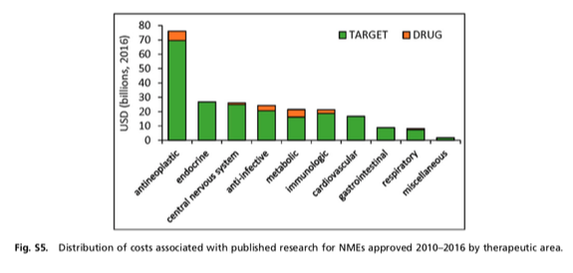
I think the quote, "The Future is already here--It's just not very evenly distributed" is most accurately attributed to William Gibson. I heard it mentioned in passing during a recent London School of Economics and Political Science podcast, Fair Shot: rethinking inequality and how we earn. Chris Hughes had truly innovative ways to rethink how we define and solve inequalities. His new book of the same name is here. Discussions of a basic income guarantee for citizens cite the Permanent Fund Dividend (PFD) paid by the state government to all citizens of Alaska. The state exploits the oil resources and everyone gets a dividend. I am curious if this might be the solution for our healthcare data. We grant access and each participant receives a percentage of the profits generated by the ginormous database. Media headlines in immuno-oncology herald novel and futuristic treatments traveling down industry pipelines to accelerated FDA approvals. But again, far fewer will benefit regardless of the enthusiasm and gargantuan funding for R&D and M&As. This doesn't mean I am against advancements in oncology--quite the opposite. I want to see the data behind the claims and curate insights based on what we know. Not what we "think" we know. I look to leaders in the field such as John Ioannidis, MD and Vinay Prasad, MD not in a sycophantic way but to pay attention to where they are pointing. Estimation of The Percentage of US Patients With Cancer Who Benefit From Genome-Driven Oncology--John Marquart, BA, Emerson Y. Chen, MD, Vinay Prasad, MD, MPHHere is a link to the article. I recommend the granularity and detail presented in the original paper. My interest is always in the data methods and statistical analyses but even if your methodology would vary, the approach is interesting and informative. The authors relied on publicly available data For each drug approval, we catalogued the name of the drug, the date of approval, the specific treatment indication, required genomic testing for that indication, the drug’s mechanism of action, relationship between the genomic aberration and drug target, and the clinical study data, specifically treatment response, per FDA drug label. My point in all this is yes, we have made technologic advances but it is only the tippy top of both bars where we can identify eligible patients followed by patients with targeted benefits. We also should be aware that 200+ new drugs approved by the FDA 2010-2016 emanated from tax payer funded research. The figure below is from Galkina Cleary et al., and demonstrates NIH funding year and costs associated with 210 new molecular entities (NME) from 2010-2016 or 151 known molecular targets for these NMEs. "The analysis shows that > 90% of this funding represents basic research related to the biological targets for drug action rather than the drugs themselves." The highest costs are for the antineoplastics as shown here. "The insights and intellectual property arising from this basic research are then transferred to the private sector for development." Here is where the pricing and access to potentially life saving drugs becomes interesting--at least from an economics perspective. If you think of medicines as a public good we would further classify them as non-rivalrous (when one person uses the public good, another can also use it) and non-excludable (costly or impossible to exclude someone from using the good).
We made health insurance a common good in 1965--think Medicare. If we had adopted the pricing mechanism attributed to Canadian Medicare at that time--It would be the equivalent of 30 trillion dollars in the pockets of US citizens. Now that would have been a more equitable future. Comments are closed.
|
Bonny is a data enthusiast applying curated analysis and visualization to persistent tensions between health policy, economics, and clinical research in oncology.
Archives
November 2020
Categories |