|
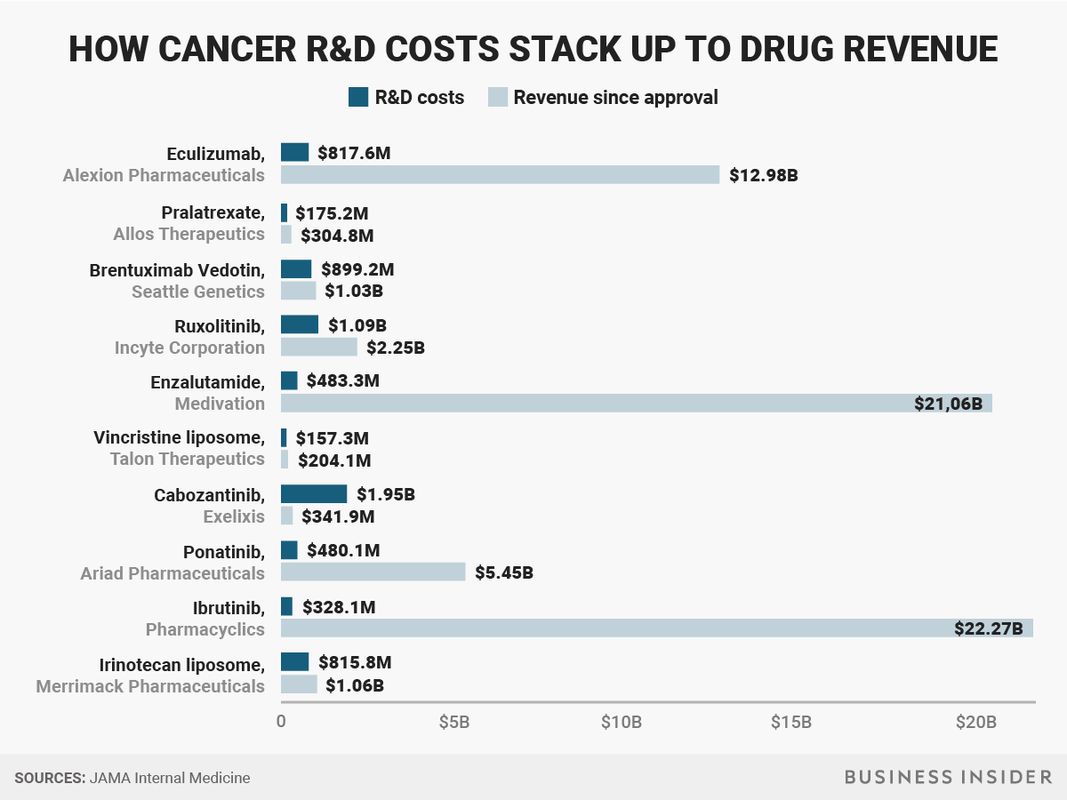
My brain hurts. Have you ever been hired as a data analyst or writer and realized perhaps the "truth" wasn't the actual requested deliverable? Maybe there are truthier truths and they are available on a sliding scale based on what pleases the client. I don't mean to be harsh but most of us have seen certain data sources preferentially requested when opposing views are found in the curated findings. Headlines often misrepresent the risks and benefits of novel therapies to the general public and clinical researchers as well. It is not a surprise--or it shouldn't be--that there is confusion at the point of care. Kite’s treatment, a form of immunotherapy called CAR-T, was initially developed by a team of researchers at the National Cancer Institute, led by a longtime friend and mentor of Dr. Belldegrun. Now Kite pays several million a year to the government to support continuing research dedicated to the company’s efforts. The relationship puts American taxpayers squarely in the middle of one of the hottest new drug markets. It also raises a question: Are taxpayers getting a good deal? “Enthusiasm for cancer immunotherapy is soaring, and so is Arie Belldegrun’s fortune.” Promising oncology drugs classified as immunotherapy are creating an effective platform for demanding top dollar. These debates all have the same framework. Research & Development costs soaring (even though M&A is hot hot hot), risky to bring drugs to market, innovation will dry up without scary pricing and looming financial profit, and a host of other reasons the market should decide who will be able to access potentially curative therapies. These are not simple tensions to resolve. What about transparency? If we are going to continue the "market" debate for pricing then we need to bring more aspects of a free market to bear. Overlooking the obvious violation of free markets (patients don't voluntarily become sick) there is a lot we don't know about the cost of producing clinical grade cellular therapies. Estimates are pretty low and vary around $14000 to $16000 per patient. But production appears to be the smallest influencer. There are supplies, QC testing, services of trained technicians, facility costs and more granular line items not described for public consumption or understanding.
The Bayh-Dole Act introduced a set of guidelines for all federal funding agencies to grant ownership of innovations to the universities and researchers that developed them. Now license terms were able to be freely negotiated leading to more ideas and economic growth. Seemingly a successful piece of legislation, Bayh-Dole secures intellectual property rights allowing commercialization of federally funded research. There has been discussion of citing the "march in" provision of the act. You may be familiar with a petition backed by Senator Bernie Sanders to allow the government to control escalating drug costs by citing the march in provision of the Bayh-Dole Act. March-in rights give the federal government the right to grant other entities licenses or even to themselves if they gained funding for the patent. The wording is being debated. Should march-in rights consider high drug costs as a threat to public safety? There are critics of a less restrictive patenting and technology transfer. The outcome--as some believe--promoted excess granting of patents and created restrictive licensing. I suggest reading the opinion rendered in Association for Molecular Pathology v. Myriad Genetics about the claim of ownership of BRCA1 and BRCA2 genes--Patents, Profits, and the American People — The Bayh–Dole Act of 1980 When the Bayh–Dole Act was written, its aim was primarily to stimulate economic growth by more efficiently mining the untapped scientific riches of hospitals, laboratories, and universities. Much has changed since then. Hard to argue that our present biotech landscape was considered back in the 1980s. How are we defining "public benefit"? Does the public as a whole benefit or are the benefits and profits funneled to a few at tax payer expense? The Bayh-Dole Act of 1980 mandates that universities take on the responsibility to transfer the novel technologies that arise from federally sponsored research. The objectives are to introduce new products in the market for public benefit and to enhance entrepreneurship--Patent and License Pearls and Pitfalls for Taking an Idea to the Marketplace I recommend the interactive calculator below for looking at value metrics around drug pricing. How much should cancer drugs cost? described the tool but the methodology is insightful as we decide how to price emerging investigational products. The Drug Pricing Lab includes the following domains allowing modifiable price components selected by user--efficacy, tolerability, novelty, research & development costs, rarity, population burden, unmet need, and prognosis. Profits and patents can be powerful incentives for scientists, businesspeople, and universities, but new and ongoing risks — including high prices that limit access to lifesaving technologies, reduced sharing of scientific data, marked shifts of focus from basic to applied research, and conflicts of interests for doctors and academic medical centers — should be mitigated or averted through revisions of the law. All Americans should be able to share in the bounties of federally funded biomedical research.--Howard Markel, MD, PhD, NEJM 2013 We are at capacity for the San Francisco venue but don't miss the all female roundtable on Facebook Live. The Future of Pharma: Getting Sh*t Done (GSD)
|
Bonny is a data enthusiast applying curated analysis and visualization to persistent tensions between health policy, economics, and clinical research in oncology.
Archives
November 2020
Categories |